Naming Aromatic Compounds Worksheet Pdf – Naming compounds is a key idea in chemistry. It involves assigning a unique name to the chemical compound on the basis of its composition. Names of compound provides important information about its properties and its structure. There are different kinds of chemical compounds, such as Ionic compounds, covalent substances, the binary type of compounds.
Naming Ionic Compounds
Ionic compounds are formed by an exchange of electrons among atoms. They are made up in positively charged caustics and negatively charged anions. The rules for naming Ionic compounds are as like this:
- Write the name for the catalytic cation in the first place, then the name of the anion.
- If the cation could have more than one possible charge Indicate the charge with Roman numerals inside parentheses.
- For anion that is not a polyatomic ion, identify the Ion.
Examples:
- NaCl is a synonym for sodium chloride.
- FeCl3 is known as iron(III) chloride.
- Mg(NO3)2 is also known as magnesium-nitrate.
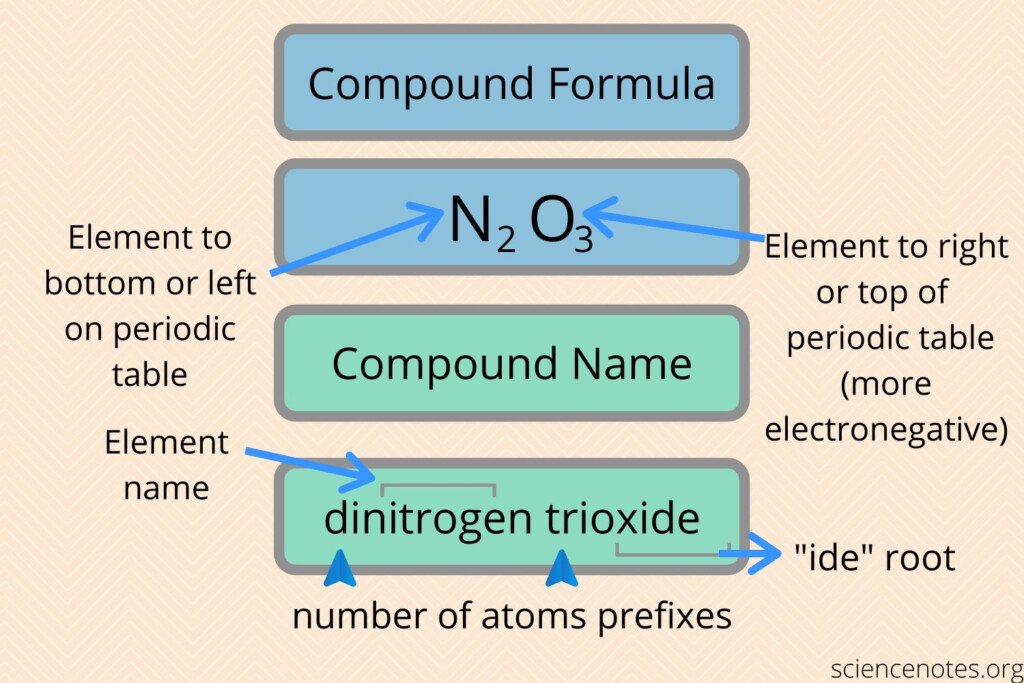
Naming Covalent Compounds
Covalent compounds are created through sharing electrons among atoms. They consist of molecules that are made by two or many atoms. The rules for naming compounds that are covalent are as follows:
- Write the name of the first element in the formula.
- Write in the first element’s name in the formula, changing the end“-ide. “-ide”.
- Use prefixes for the number of atoms for every element of the molecule. Except for the prefix “mono-” for the first element.
Examples:
- CO2 is also known as carbon dioxide.
- N2O is named dinitrogen monoxide.
- This is known as sulfur hexafluoride.
Naming Binary Compounds
Binary compounds are compounds made up of two elements. The rules for naming binary compounds are as the following:
- Write the name of the first element of the formula.
- Write in the first element’s name in the formula, changing the end“-ide” to “-ide”.
Examples:
- Hydrogen chloride is also known as hydrogen.
- CO is named carbon monoxide.
- CaO is named calcium oxide.
Practice Exercises
For reinforcement of learning The worksheet will provide activities for practicing naming ionic components, covalent compounds, or binary substances. This will help students establish a firm understanding of what rules are used for naming chemical compounds.
Ionic Compound Naming Exercises:
- Na2S
- KBr
- CaF2
- Al2O3
Covalent Compound Naming Exercises:
- CO
- SO2
- N2O4
- H2O2
Binary Compound Naming Exercises:
- Cl2O7
- P2S5
- BrF3
- NO
If they can complete these assignments, learners will become confident in the identification of chemical compounds, and will be able to apply these rules to other chemical compounds.
Conclusion:
Naming compounds is a crucial concept in chemistry that requires a clear understanding of fundamental rules and principles to naming different types of compounds. In following the principles laid out in this worksheet and practicing using the provided exercises, students will be able confidently identify ionic and covalent along with binary and covalent compounds. This knowledge is crucial for an effective chemistry education and forms the foundation for further research in the area.